Abstract
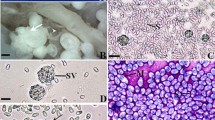
A novel microsporidial disease was documented in two ornamental fish species, black tetra Gymnocorymbus ternetzi Boulenger 1895 and cardinal tetra Paracheirodon axelrodi Schultz 1956. The non-xenoma-forming microsporidium occurred diffusely in most internal organs and the gill, thus referring to the condition as tetra disseminated microsporidiosis (TDM). The occurrence of TDM in black tetra was associated with chronic mortality in a domestic farmed population, while the case in cardinal tetra occurred in moribund fish while in quarantine at a public aquarium. Histology showed that coelomic visceral organs were frequently necrotic and severely disrupted by extensive infiltrates of macrophages. Infected macrophages were presumed responsible for the dissemination of spores throughout the body. Ultrastructural characteristics of the parasite developmental cycle included uninucleate meronts directly in the host cell cytoplasm. Sporonts were bi-nucleated as a result of karyokinesis and a parasite-produced sporophorous vesicle (SPV) became apparent at this stage. Cytokinesis resulted in two spores forming within each SPV. Spores were uniform in size, measuring about 3.9 ± 0.33 long by 2.0 ± 0.2 μm wide. Ultrastructure demonstrated two spore types, one with 9–12 polar filament coils and a double-layered exospore and a second type with 4–7 polar filament coils and a homogenously electron-dense exospore, with differences perhaps related to parasite transmission mechanisms. The 16S rDNA sequences showed closest identity to the genus Glugea (≈ 92%), though the developmental cycle, specifically being a non-xenoma-forming species and having two spores forming within a SPV, did not fit within the genus. Based on combined phylogenetic and ultrastructural characteristics, a new genus (Fusasporis) is proposed, with F. stethaprioni n. gen. n. sp. as the type species.







Similar content being viewed by others
Data availability
Histology slides were submitted to the National Parasite Collection housed at the Smithsonian Institution, National Museum of Natural History, Department of Invertebrate Zoology, under catalog numbers USNM1638543–USNM1638546. Genetic sequence of the small subunit rDNA was deposited to GenBank under accession number MW077214. Original histology blocks and resin-embedded blocks are maintained at the Tropical Aquaculture Laboratory, University of Florida and the Office of Fish and Wildlife Health and Forensics Laboratory, NJ Division of Fish and Wildlife, respectively.
References
Abdel-Baki A-AS, Al-Quraishy S, Rocha S, Dkhil MA, Casal G, Azevedo C (2015a) Ultrastructure and phylogeny of Glugea nagelia sp. n. (Microsporidia: Glugeidae), infecting the intestinal wall of the yellowfin hind, Cephalopholis hemistiktos (Actinopterygii: Serranidae), from the Red Sea. Folia Parasitol (Phraha) 62. https://doi.org/10.14411/fp.2015.007
Abdel-Baki A-AS, Tamihi AF, Al-Qahtani HA, Al-Quraishy S, Mansour L (2015b) Glugea jazanensis sp. nov. infecting Lutjanus bohar in the Red Sea: ultrastructure and phylogeny. Dis Aquat Org 116:185–190. https://doi.org/10.3354/dao02927
Abdel-Baki AS, Al-Quraishy S, Al-Qahtani H, Dkhil MA, Azevedo C (2012) Morphological and ultrastructural description of Pleistophora dammami sp. n. infecting the intestinal wall of Saurida undosquamis from the Arabian Gulf, Saudi Arabia. Parasitol Res 111:413–418. https://doi.org/10.1007/s00436-012-2855-4
Al-Quraishy S, Abdel-Baki AS, Al-Qahtani H, Dkhil M, Casal G, Azevedo C (2012) A new microsporidian parasite, Heterosporis saurida n. sp. (Microsporidia) infecting the lizardfish, Saurida undosquamis from the Arabian Gulf, Saudi Arabia: ultrastructure and phylogeny. Parasitology 139:454–462. https://doi.org/10.1017/S0031182011001971
Azevedo C, Abdel-Baki A-AS, Rocha S, Al-Quraishy S, Casal G (2016) Ultrastructure and phylogeny of Glugea arabica n. sp. (Microsporidia), infecting the marine fish Epinephelus polyphekadion from the Red Sea. Eur J Protistol 52:11–21. https://doi.org/10.1016/j.ejop.2015.09.003
Bader JA, Shotts EB, Steffens WL, Lom J (1998) Occurrence of Loma cf. salmonae in brook, brown and rainbow trout from Buford trout hatchery, Georgia, USA. Dis Aquat Org 34:211–216. https://doi.org/10.3354/dao034211
Bass D, Czech L, Williams BAP, Berney C, Dunthorn M (2018) Clarifying the relationships between Microsporidia and Cryptomycota. J Eukaryot Microbiol 65:773–782. https://doi.org/10.1111/jeu.12519
Benine RC, Melo BF, Castro RMC, Oliveira C (2015) Taxonomic revision and molecular phylogeny of Gymnocorymbus Eigenmann, 1908 (Teleostei, Characiformes, Characidae). Zootaxa 3956:1–28. https://doi.org/10.11646/zootaxa.3956.1.1
Bigliardi E, Selmi MG, Lupetti P, Corona S, Gatti S, Scaglia M, Sacchi L (1996) Microsporidian spore wall: ultrastructural findings on Encephalitozoon hellem exospore. J Eukaryot Microbiol 43:181–186. https://doi.org/10.1111/j.1550-7408.1996.tb01388.x
Brown AMV, Kent ML, Adamson ML (2010a) Description of five new Loma (Microsporidia) species in Pacific fishes with redesignation of the type species Loma morhua Morrison & Sprague, 1981, based on morphological and molecular species-boundaries tests. J Eukaryot Microbiol 57:529–553. https://doi.org/10.1111/j.1550-7408.2010.00508.x
Brown AMV, Kent ML, Adamson ML (2010b) Low genetic variation in the salmon and trout parasite Loma salmonae (Microsporidia) supports marine transmission and clarifies species boundaries. Dis Aquat Org 91:35–46. https://doi.org/10.3354/dao02246
Cali A, Takvorian PM (2014) Developmental morphology and life cycles of the Microsporidia. In: Weiss LM, Becnel JJ (eds) Microsporidia pathogens of opportunity, 1st edn. Wiley Blackwell, Ames, pp 71–134
Camus AC, Dill JA, Rosser TG, Pote LM, Griffin MJ (2017) Myxobolus axelrodi n. sp. (Myxosporea: Myxobolidae) a parasite infecting the brain and retinas of the cardinal tetra Paracheirodon axelrodi (Teleostei: Characidae). Parasitol Res 116:387–397. https://doi.org/10.1007/s00436-016-5301-1
Canning EU, Nicholas JP (1980) Genus Pleistophora (phylum Microspora): redescription of the type species, Pleistophora typicalis Gurley, 1893 and ultrastructural characterization of the genus. J Fish Dis 3:317–338. https://doi.org/10.1111/j.1365-2761.1980.tb00402.x
Casal G, Matos E, Teles-Grilo ML, Azevedo C (2009) Morphological and genetical description of Loma psittaca sp. n. isolated from the Amazonian fish species Colomesus psittacus. Parasitol Res 105:1261–1271. https://doi.org/10.1007/s00436-009-1547-1
Casal G, Rocha S, Costa G, Al-Quraishy S, Azevedo C (2016) Ultrastructural and molecular characterization of Glugea serranus n. sp., a microsporidian infecting the blacktail comber, Serranus atricauda (Teleostei: Serranidae), in the Madeira archipelago (Portugal). Parasitol Res 115:3963–3972. https://doi.org/10.1007/s00436-016-5162-7
Costa G, Melo-Moreira E, de Carvalho MAP (2016) Occurrence of microsporidians Glugea hertwigi and Pleistophora ladogensis, in smelt Osmerus eperlanus from two German rivers, North Sea coast. Dis Aquat Org 121:49–57. https://doi.org/10.3354/dao03040
Couch JA (1978) Diseases, parasites, and toxic responses of commercial penaeid shrimps of the Gulf of Mexico and South Atlantic coasts of North America. Fish Bull 76:1–44
Dezfuli BS, Giari L, Simoni E, Shinn AP, Bosi G (2004) Immunohistochemistry, histopathology and ultrastructure of Gasterosteus aculeatus tissues infected with Glugea anomala. Dis Aquat Org 58:193–202. https://doi.org/10.3354/dao058193
Didier ES (1997) Effects of albendazole, fumagillin, and TNP-470 on microsporidial replication in vitro. Antimicrob Agents Chemother 41:1541–1546
El-Garhy M, Cali A, Morsy K, Bashtar AR, Al-Quraishy S (2017) Ultrastructural characterization of Pleistophora macrozoarcidis Nigerelli 1946 (Microsporidia) infecting the ocean pout Macrozoarces americanus (Perciformes, Zoarcidae) from the gulf of Maine, MA, USA. Parasitol Res 116:61–71. https://doi.org/10.1007/s00436-016-5261-5
Evers H-G, Pinnegar JK, Taylor MI (2019) Where are they all from? – sources and sustainability in the ornamental freshwater fish trade. J Fish Biol 94:909–916. https://doi.org/10.1111/jfb.13930
Ferguson JA, Watral V, Schwindt AR, Kent ML (2007) Spores of two fish microsporidia (Pseudoloma neurophilia and Glugea anomala) are highly resistant to chlorine. Dis Aquat Org 76:205–214. https://doi.org/10.3354/dao076205
Filleul A, Maisey JG (2004) Redescription of Santanichthys diasii (Otophysi, Characiformes) from the Albian of the Santana Formation and comments on its implications for otophysan relationships. Am Mus Novit 2004:1–21. https://doi.org/10.1206/0003-0082(2004)455<0001:ROSDOC>2.0.CO;2
Géry J (1977) Characoids of the world. Tropical Fish Hobbyist Publications, Inc., Neptune City
Goertz D, Hoch G (2008) Horizontal transmission pathways of terrestrial microsporidia: a quantitative comparison of three pathogens infecting different organs in Lymantria dispar L. (Lep.: Lymantriidae) larvae. Biol Control 44:196–206. https://doi.org/10.1016/j.biocontrol.2007.07.014
Han B, Weiss LM (2017) Microsporidia: obligate intracellular pathogens within the fungal kingdom. Microbiol Spectr 5. https://doi.org/10.1128/microbiolspec.FUNK-0018-2016
Hanaichi T, Sato T, Iwamoto T, Malavasi-Yamashiro J, Hoshino M, Mizuno N (1986) A stable lead by modification of Sato’s method. J Electron Microsc 35:304–306. https://doi.org/10.1093/oxfordjournals.jmicro.a050582
Higes M, Nozal MJ, Alvaro A, Barrios L, Meana A, Martín-Hernández R, Bernal JL, Bernal J (2011) The stability and effectiveness of fumagillin in controlling Nosema ceranae (Microsporidia) infection in honey bees (Apis mellifera) under laboratory and field conditions. Apidologie 42:364–377. https://doi.org/10.1007/s13592-011-0003-2
Huang WF, Solter LF, Yau PM, Imai BS (2013) Nosema ceranae escapes fumagillin control in honey bees. PLoS Pathog 9:e1003185. https://doi.org/10.1371/journal.ppat.1003185
Joh SJ, Kwon YK, Kim MC, Kim MJ, Kwon HM, Park JW, Kwon JH, Kim JH (2007) Heterosporis anguillarum infections in farm cultured eels (Anguilla japonica) in Korea. J Vet Sci 8:147–149. https://doi.org/10.4142/jvs.2007.8.2.147
Kent ML, Buchner C, Watral VG, Sanders JL, LaDu J, Peterson TS, Tanguay RL (2011) Development and maintenance of a specific pathogen free (SPF) zebrafish research facility for Pseudoloma neurophilia. Dis Aquat Org 95:73–79. https://doi.org/10.3354/dao02333
Kent ML, Shaw RW, Sanders JL (2014) Microsporidia in fish. In: Weiss LM, Becnel JJ (eds) Microsporidia pathogens of opportunity, 1st edn. Ames IA, Wiley Blackwell, pp 493–520
Khan RA (2005) Prevalence and influence of Loma branchialis (Microspora) on growth and mortality in Atlantic cod (Gadus morhua) in coastal Newfoundland. J Parasitol 91:1230–1232. https://doi.org/10.1645/GE-528R.1
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Lee SC, Corradi N, Byrnes EJ, Torres-Martinez S, Dietrich FS, Keeling PJ, Heitman J (2008) Microsporidia evolved from ancestral sexual fungi. Curr Biol CB 18:1675–1679. https://doi.org/10.1016/j.cub.2008.09.030
Li K, Chang O, Wang F, Liu C, Liang H, Wu S (2012) Ultrastructure, development, and molecular phylogeny of Pleistophora hyphessobryconis, a broad host microsporidian parasite of Puntius tetrazona. Parasitol Res 111:1715–1724. https://doi.org/10.1007/s00436-012-3013-8
Lom J, Dykova I (2005) Microsporidian xenomas in fish seen in wider perspective. Folia Parasitol 52:69–81. https://doi.org/10.14411/fp.2005.010
Lom J, Dyková I, Wang CH, Lo CF, Kou GH (2000) Ultrastructural justification for the transfer of Pleistophora anguillarum Hoshina, 1959 to the genus Heterosporis Schubert, 1969. Dis Aquat Org 43:225–231. https://doi.org/10.3354/dao043225
Lom J, Nilsen F (2003) Fish microsporidia: fine structural diversity and phylogeny. Int J Parasitol 33:107–127. https://doi.org/10.1016/s0020-7519(02)00252-7
Lom J, Noga E, Dyková I (1995) Occurrence of a microsporean with characteristics of Glugea anomala in ornamental fish of the family Cyprinodontidae. Dis Aquat Org 21:239–242. https://doi.org/10.3354/dao021239
Lovy J, Friend SE (2017) Phylogeny and morphology of Ovipleistophora diplostomuri n. sp. (Microsporidia) with a unique dual-host tropism for bluegill sunfish and the digenean parasite Posthodiplostomum minimum (Strigeatida). Parasitology 144:1898–1911. https://doi.org/10.1017/S0031182017001305
Lovy J, Kostka M, Dyková I, Arsenault G, Pecková H, Wright GM, Speare DJ (2009) Phylogeny and morphology of Glugea hertwigi from rainbow smelt Osmerus mordax found in Prince Edward Island, Canada. Dis Aquat Org 86:235–243. https://doi.org/10.3354/dao02133
Lovy J, Wadowska DW, Wright GM, Speare DJ (2004) Morphological characterization and notes on the life cycle of a newly discovered variant of Loma salmonae (Putz, Hoffman & Dunbar) from a natural infection of Chinook salmon, Oncorhynchus tshawytscha (Walbaum). J Fish Dis 27:609–616. https://doi.org/10.1111/j.1365-2761.2004.00582.x
Luna LG, Armed Forces Institute of Pathology (U.S.), Armed Forces Institute of Pathology (U.S.) (1968) Manual of histologic staining methods of the Armed Forces Institute of Pathology. Blakiston Division, McGraw-Hill
Mansour L, Zhang JY, Abdel-Haleem HM, Darwish AB, Al-Quraishy S, Abdel-Baki AA (2020) Ultrastructural description and phylogeny of a novel microsporidian, Glugea eda n. sp. from the striated fusilier, Caesio striata, in the Red Sea off Saudi Arabia. Acta Trop 204:105331. https://doi.org/10.1016/j.actatropica.2020.105331
Matthews JL, Brown AM, Larison K, Bishop-Stewart JK, Rogers P, Kent ML (2001) Pseudoloma neurophilia n. g., n. sp., a new microsporidium from the central nervous system of the zebrafish (Danio rerio). J Eukaryot Microbiol 48:227–233. https://doi.org/10.1111/j.1550-7408.2001.tb00307.x
Maurand J, Loubes C, Gasc C, Pelletier J, Barral J (1988) Pleistophora mirandellae Vaney & Conte, 1901, a microsporidian parasite in cyprinid fish of rivers in Herault: taxonomy and histopathology. J Fish Dis 11:251–258. https://doi.org/10.1111/j.1365-2761.1988.tb00546.x
Moore CB, Brooks WM (1992) An ultrastructural study of Vairimorpha necatrix (Microspora, Microsporida) with particular reference to episporontal inclusions during octosporogony. J Protozool 39:392–398. https://doi.org/10.1111/j.1550-7408.1992.tb01469.x
Morsy K, Abdel-Ghaffar F, Mehlhorn H, Bashtar AR, Abdel-Gaber R (2012) Ultrastructure and molecular phylogenetics of a new isolate of Pleistophora pagri sp. nov. (Microsporidia, Pleistophoridae) from Pagrus pagrus in Egypt. Parasitol Res 111:1587–1597. https://doi.org/10.1007/s00436-012-3012-9
Nei M, Kumar S (2000) Molecular evolution and phylogenetics. Oxford University Press, New York
Nilsen F, Endresen C, Hordvik I (1998) Molecular phylogeny of microsporidians with particular reference to species that infect the muscles of fish. J Eukaryot Microbiol 45:535–543. https://doi.org/10.1111/j.1550-7408.1998.tb05113.x
Oliveira C, Avelino GS, Abe KT, Mariguela TC, Benine RC, Orti G, Vari RP, Corrêa e Castro R (2011) Phylogenetic relationships within the speciose family Characidae (Teleostei: Ostariophysi: Characiformes) based on multilocus analysis and extensive in group sampling. BMC Evol Biol 11:275. https://doi.org/10.1186/1471-2148-11-275
Ovcharenko M, Wróblewski P, Kvach Y, Drobiniak O (2017) Study of Loma acerinae (Microsporidia) detected from three Ponto-Caspian gobies (Gobiidae) in Ukraine. Parasitol Res 116:1453–1462. https://doi.org/10.1007/s00436-017-5422-1
Pekkarinen M, Lom J, Nilsen F (2002) Ovipleistophora gen. n., a new genus for Pleistophora mirandellae-like microsporidia. Dis Aquat Org 48:133–142. https://doi.org/10.3354/dao048133
Phelps NBD, Goodwin AE (2008) Vertical transmission of Ovipleistophora ovariae (microspora) within the eggs of the golden shiner. J Aquat Anim Health 20:45–53. https://doi.org/10.1577/H07-029.1
Phelps NBD, Mor SK, Armién AG, Pelican KM, Goyal SM (2015) Description of the microsporidian parasite, Heterosporis sutherlandae n. sp., infecting fish in the Great Lakes region, USA. PLoS One 10:e0132027. https://doi.org/10.1371/journal.pone.0132027
Pomport-Castillon C, De Jonckheere JF, Romestand B (2000) Ribosomal DNA sequences of Glugea anomala, G. stephani, G. americanus and Spraguea lophii (Microsporidia): phylogenetic reconstruction. Dis Aquat Org 40:125–129. https://doi.org/10.3354/dao040125
Raghavan R, Dahanukar N, Tlusty MF, Rhyne AL, Krishna KK, Molur S, Rosser AM (2013) Uncovering an obscure trade: threatened freshwater fishes and the aquarium pet markets. Biol Conserv 164:158–169. https://doi.org/10.1016/j.biocon.2013.04.019
Rodriguez-Tovar LE, Wadowska DW, Wright GM, Groman DB, Speare DJ, Whelan DS (2003) Ultrastructural evidence of autoinfection in the gills of Atlantic cod Gadus morhua infected with Loma sp. (phylum Microsporidia). Dis Aquat Org 57:227–230. https://doi.org/10.3354/dao057227
Rodríguez-Tovar LE, Wright GM, Wadowska DW, Speare DJ, Markham RJF (2003) Ultrastructural study of the late stages of Loma salmonae development in the gills of experimentally infected rainbow trout. J Parasitol 89:464–474. https://doi.org/10.1645/0022-3395(2003)089[0464:USOTLS]2.0.CO;2
Ryan JA, Kohler SL (2016) Distribution, prevalence, and pathology of a microsporidian infecting freshwater sculpins. Dis Aquat Org 118:195–206. https://doi.org/10.3354/dao02974
Sanders J, Myers MS, Tomanek L, Cali A, Takvorian PM, Kent ML (2012) Ichthyosporidium weissii n. sp. (Microsporidia) infecting the arrow goby (Clevelandia ios). J Eukaryot Microbiol 59:258–267. https://doi.org/10.1111/j.1550-7408.2012.00619.x
Sanders JL, Lawrence C, Nichols DK, Brubaker JF, Peterson TS, Murray KN, Kent ML (2010) Pleistophora hyphessobryconis (Microsporidia) infecting zebrafish Danio rerio in research facilities. Dis Aquat Org 91:47–56. https://doi.org/10.3354/dao02245
Sanders JL, Watral V, Stidworthy MF, Kent ML (2016) Expansion of the known host range of the microsporidium, Pseudoloma neurophilia. Zebrafish 13:S102–S106. https://doi.org/10.1089/zeb.2015.1214
Solter LF, Maddox JV (1998) Timing of an early sporulation sequence of microsporidia in the genus Vairimorpha (Microsporidia: Burenellidae). J Invertebr Pathol 72:323–329. https://doi.org/10.1006/jipa.1998.4815
Speare DJ, Arsenault G, Buote M (1998) Evaluation of rainbow trout as a model for use in studies on pathogenesis of the branchial Microsporidian Loma salmonae. Contemp Top Lab Anim Sci 37:55–58
Speare DJ, Athanassopoulou F, Daley J, Sanchez JG (1999) A preliminary investigation of alternatives to fumagillin for the treatment of Loma salmonae infection in rainbow trout. J Comp Pathol 121:241–248. https://doi.org/10.1053/jcpa.1999.0325
Steffens W (1962) Der Heutige Stand Der Verbreitung Von Pliestophora hyphessobryconis Schäperclaus 1941 (Sporozoa, Microsporidia). Z Für Parasitenkd 21:535–541. https://doi.org/10.1007/BF00260258
Su Y, Feng J, Sun X, Jiang J, Guo Z, Ye L, Xu L (2014) A new species of Glugea Thélohan, 1891 in the red sea bream Pagrus major (Temminck & Schlegel) (Teleostei: Sparidae) from China. Syst Parasitol 89:175–183. https://doi.org/10.1007/s11230-014-9519-y
Takvorian PM, Cali A (1983) Appendages associated with Glugea stephani, a microsporidan found in flounder. J Protozool 30:251–256. https://doi.org/10.1111/j.1550-7408.1983.tb02911.x
Vagelli A, Paramá A, Sanmartín ML, Leiro J (2005) Glugea vincentiae n. sp. (Microsporidia: Glugeidae) infecting the Australian marine fish Vincentia conspersa (Teleostei: Apogonidae). J Parasitol 91:152–157. https://doi.org/10.1645/GE-388R
Vávra J, Larsson JIR (2014) Structure of Microsporidia. In: Weiss LM, Becnel JJ (eds) Microsporida pathogens of opportunity, 1st edn. Wiley Blackwell, Ames, pp 1–70
Voronin VN (1976) Characteristics of the genus Glugea (Protozoa, Microsporidia) based on the example of the type species Glugea anomala (Moniez, 1887) Gurley 1893 and its varieties. Parazitologiia 10:263–267
Vossbrinck CR, Andreadis TG, Vavra J, Becnel JJ (2004) Molecular phylogeny and evolution of mosquito parasitic microsporidia (Microsporidia: Amblyosporidae). J Eukaryot Microbiol 51:88–95. https://doi.org/10.1111/j.1550-7408.2004.tb00167.x
Vossbrinck CR, Baker MD, Didier ES, Debrunner-Vossbrinck BA, Shadduck JA (1993) Ribosomal DNA sequences of Encephalitozoon hellem and Encephalitozoon cuniculi: species identification and phylogenetic construction. J Eukaryot Microbiol 40:354–362. https://doi.org/10.1111/j.1550-7408.1993.tb04928.x
Vossbrinck CR, Debrunner-Vossbrinck BA (2005) Molecular phylogeny of the Microsporidia: ecological, ultrastructural and taxonomic considerations. Folia Parasitol 52:131–142. https://doi.org/10.14411/fp.2005.017
Weidner E (1972) Ultrastructural study of microsporidian invasion into cells. Z Für Parasitenkd 40:227–242. https://doi.org/10.1007/BF00329623
Winters AD, Langohr IM, Souza MDEA, Colodel EM, Soares MP, Faisal M (2016) Ultrastructure and molecular phylogeny of Pleistophora hyphessobryconis (Microsporidia) infecting hybrid jundiara (Leiarius marmoratus × Pseudoplatystoma reticulatum) in a Brazilian aquaculture facility. Parasitol 143:41–49. https://doi.org/10.1017/S0031182015001420
Zehev BS, Vera A, Asher B, Raimundo R (2015) Ornamental fishery in Rio Negro (Amazon region), Brazil: combining social, economic and fishery analyses. https://doi.org/10.4172/2150-3508.1000143
Acknowledgments
The authors thank John Leary and Dr. Lowia Al-Hussinee for assistance with processing samples for molecular analyses.
Funding
This study was funded by combined internal funds from the authors’ host laboratories.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
Not applicable.
Consent for participation and publication
All authors have provided consent for participation and publication of this study.
Additional information
Section Editor: Shokoofeh Shamsi
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 436 kb)
Rights and permissions
About this article
Cite this article
Lovy, J., Yanong, R.P.E., Stilwell, J.M. et al. Tetra disseminated microsporidiosis: a novel disease in ornamental fish caused by Fusasporis stethaprioni n. gen. n. sp.. Parasitol Res 120, 497–514 (2021). https://doi.org/10.1007/s00436-020-06988-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-020-06988-7